QCM-D is a versatile and powerful technique that is widely used in biointerface science and in research areas such as biophysics, biotechnology, and biomedical engineering. The method provides real-time, high-sensitivity measurements of mass changes and viscoelastic properties of thin layers at the sensor surface and is used to study both biomolecular interactions and interactions at solid-liquid interfaces. We have compiled a brief overview that describes how QSense QCM-D can be used to analyze biomolecular interactions and what information QCM-D measurements offer
QCM-D is a surface sensitive technology which has been used to analyze biomolecular interactions for two decades. Via time-resolved information on mass, thickness and viscoelastic properties of surface adhering layers, the method can detect and monitor molecular interactions and events in real-time. Interactions and interfacial processes that can be analyzed are for example:
QCM-D measures the so called “hydrated mass”. This feature makes it an excellent complement to optical time-resolved technologies, which are often used to study biomolecular interactions, and which senses “non-hydrated mass”. Whereas the non-hydrated mass refers to the mass of the biomolecules, the hydrated mass includes both the molecules and the surrounding solvent. Monitoring the hydrated mass enables not only detection of surface interactions such as adsorption, desorption, and binding, but it also enables the detection of molecular arrangement at the surface and changes thereof. Fig. 1 illustrates what typical data could look like in the cases of A) adsorption, B) binding and C) enzymatic action.
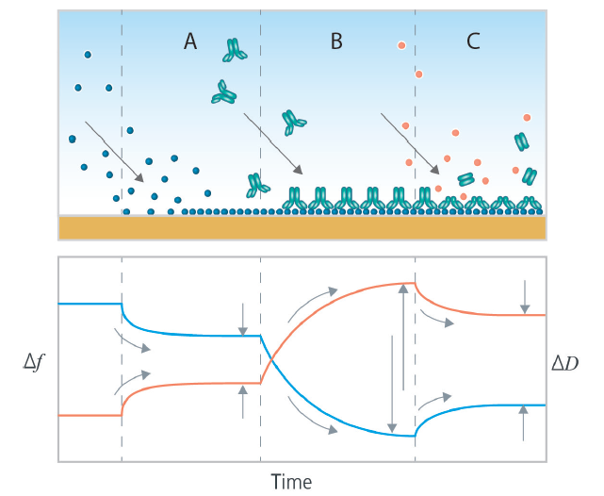
Figure 1. Schematic illustration (top panel) of (A) molecular adsorption, (B) binding and (C) enzymatic action, characterized by QSense QCM-D (middle panel). The Δf and ΔD data reflect time-resolved mass uptake and layer softness respectively. As indicated by the grey arrows in the schematic graphs, the time-resolved data makes it possible to follow the adsorption, binding and enzymatic action and analyze how fast they are, and how much material that is added to or lost from the surface in the respective process. The absolute amount adsorbed, bound and enzymatically removed can be also analyzed via quantification of the time-resolved layer thickness or mass (not shown).
Running QCM-D analysis at relevant conditions and varying, for example, the surface material, the temperature, the pH, or the salt concentration, the behavior of the biomolecular system can be both characterized for deeper understanding and optimized for a target application. Questions that can be answered are for example:
Download the overview to read more about how biomolecular-based systems can be characterized using QSense QCM-D technology and to get examples of what typical data could look like in the different cases.
Editor’s note: This post was originally published in Dec 2019 and has been updated.
Learn about how QSense analysis can be used to assess adsorption of biologics and excipients and reduce the risk of late discoveries of incompatibilities
Read about how protein adsorption at various surface and solution conditions quickly can be measured
QCM-D is utilized to understand the compatibility and to identify solution and excipient conditions that minimize the adsorption of therapeutic proteins
Read about how QSense QCM-D analysis is used in the quest to tackle inflammation and bacterial infections on implant surfaces.
Learn more about what questions QSense analysis can help answer in the context of container-closure interaction
Recent research results could help designing formulations better stabilized against protein aggregation, one of the main challenges in pharmaceutical development. QSense QCM-D analysis helped piecing the puzzle together
Read about how Dr. Gustaf Rydell, researcher at Sahlgrenska University Hospital, uses QCM-D to understand how the Norovirus initiates infection.
Here we present a list of scientific publications in virus-related research where QCM-D technology has been used
