
Polymers and polyelectrolytes of various conformations are used in many applications where there is a need to tailor the interfacial properties to promote a certain interaction with the surrounding environment. Here we show how polymer layer crosslinking and collapse can be characterized.
To tailor the interfacial properties of these layers, it is important to characterize and understand the conformational behavior, such as the degree of hydration and transitions from a hydrated to a collapsed or crosslinked state, and vice versa, Figure 1. The swelling and collapse of polymer brushes, and other thin films, can be characterized by QCM-D and other technologies, which will sense the water uptake and water release as changes in mass.
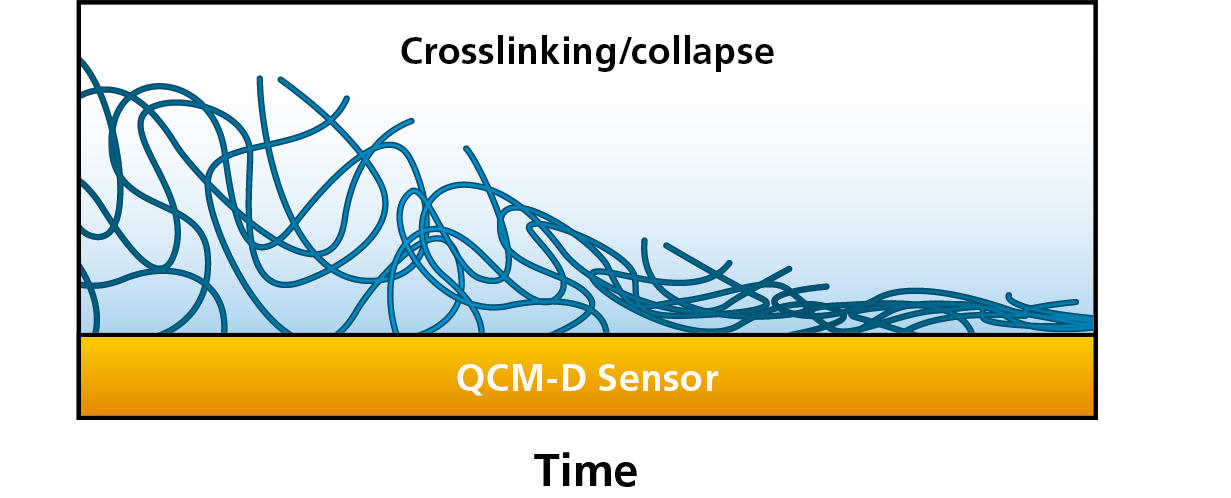
Figure 1. From left to right, this schematic illustration shows how a thick and hydrated film releases water and collapses into a thin layer at the surface.
As an example, let’s look at the transitions between hydrated and dehydrated states of polymer brushes made of chitosan. At low pH, the brush will be in a hydrated state, while at high pH it will be in a dehydrated state. It is also possible to use anions for crosslinking of the brush.
First, the QCM-D sensor is coated with the polymer. Next, the chitosan polymer brush layer is exposed to solutions of different pH and counter-anion type and the effect on the film thickness is monitored.
The results, Fig. 2., show how the thickness of the polymer varies as it goes through the swelling and collapse transformation when the pH and counter-anion are varied. It is also interesting to note that when the citrate anions are replacing the acetate anions (similar pH, step 2 in the figure), ionic cross-links are formed, which results in a collapse of brush layer.
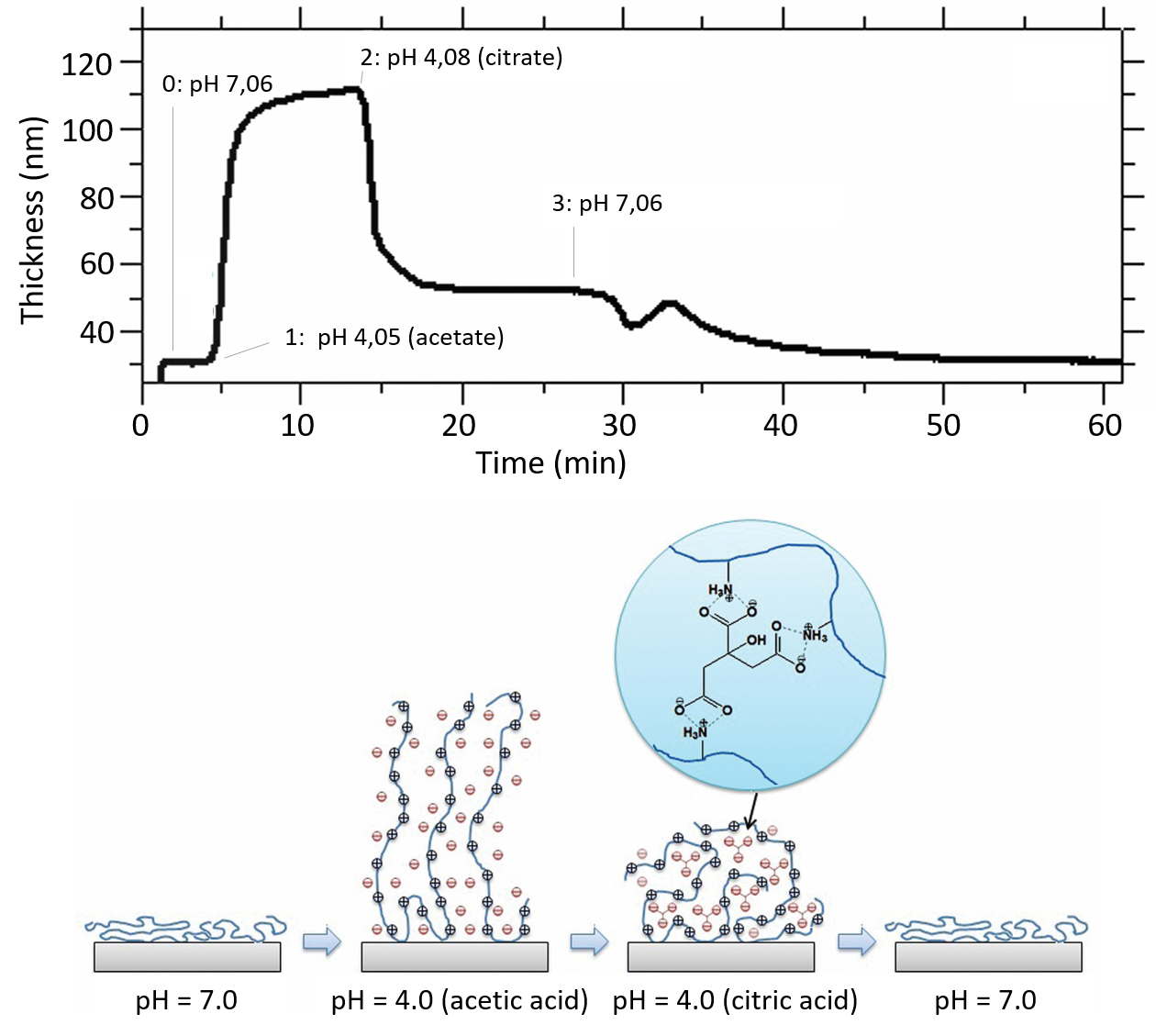
Figure 21. (Top) The thickness of the Chitosan brush layers when exposed to different solution pH and counter-ions. (Bottom) Schematic illustration of the structure of the chitosan brush layer as a function of pH and counter-ion type.
Polymer brushes, multilayers and hydrogels, i.e. cross-linked polymer networks, are all, more or less, hydrated and viscoelastic, depending on the molecular conformation at the interface. The conformation will have a major impact on the resulting interfacial properties and will influence the interaction with the ambient such as e.g. protein adsorption or prevention of bacteria adhesion. The conformation of the polymer layer can be characterized with for example QCM-D, which also enables straightforward detection of polymer brush transitions between hydrated and non-hydrated states.
Download the overview to read more about what information you can obtain with QSense QCM-D.
1. H-S Lee, et al., J. Mater. Chem., 22, 19605, 2012
Register for the webinar
Read the guidelines on how to decide which QCM instrument will be the most suitable for your needs
Read about how protein adsorption at various surface and solution conditions quickly can be measured
Read about what single-harmonic and multi-harmonic QCM-D means and what the difference is between these instruments.
Read about how QSense QCM-D analysis is used as a powerful tool to investigate protein-lipid nanoparticles binding affinity
Learn about the difference between the theoretical QCM sensitivity and the sensitivity which is relevant in a measurement situation.
Read about the piezoelectric effect and how piezoelectricity arises
Learn more about QSense 4th generation QCM-D platform which provides a sharper tool in the scientist toolbox and simplifies data interpretation.
Learn about how biointerfaces and biomolecular interactions can be studied using QSense® QCM-D and what information these measurements offer.
Read about Prof. Jackman's experience using QCM-D to study surfactant-interaction with model membranes
Sign upp for the webinar to learn more about how QCM-D is used to study biomaterial-induced activation of the immune system
Gabriel Ohlsson, Ph.D., is a former employee at Biolin Scientific where he initially held a position as an application scientist and later as a sales manager. Dr. Ohlsson did his Ph.D. in engineering physics and has spent a lot of time developing sensing technologies for soft matter material applications. One of his main tools during this research has been the QCM-D technology.
