Surfactants are the most versatile products of the chemical industry. 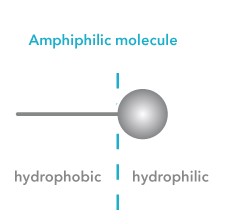 They are utilized in every industrial area ranging from household detergents to drilling muds and food items to pharmaceuticals.
They are utilized in every industrial area ranging from household detergents to drilling muds and food items to pharmaceuticals.
The term surfactant comes from the word surface active agent. They are amphiphilic molecules and are thus absorbed in the air-water interface. At the interface, they align themselves so that the hydrophobic part is in the air and the hydrophilic part is in water. This will cause a decrease in surface or interfacial tensions.
Surfactant basics
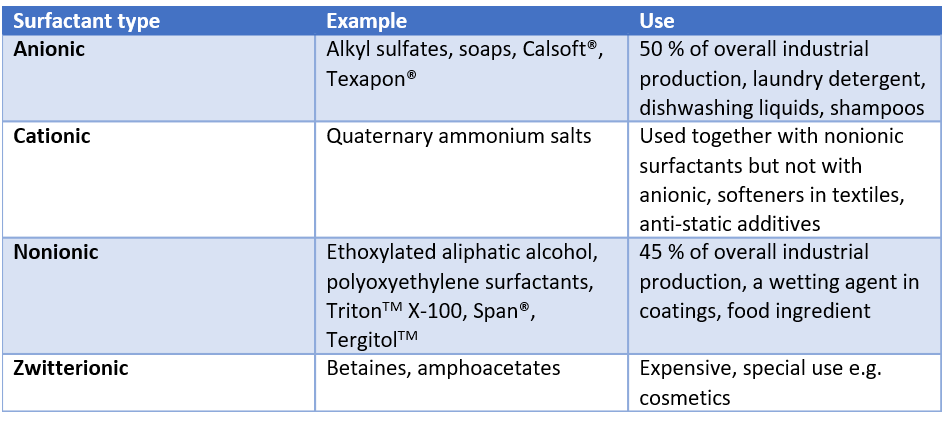
As said, surfactants are amphiphilic molecules that have hydrophobic and hydrophilic parts. The hydrophobic tail is a hydrocarbon, fluorocarbon, or siloxane. Surfactants are typically classified based on their polar head as the hydrophobic tails are often similar. If the head group has no charge, the surfactant is called non-ionic. If the head group has a negative or positive charge, it is called anionic or cationic, respectively. If it contains both positive and negative groups, then the surfactant is called zwitterionic.
Anionic and nonionic surfactants are by far the most used surfactant types in the industry. Anionic surfactant finds use, especially in cleaning products like laundry detergents and shampoos. Nonionic surfactants on the other hand are often used as wetting agents and in the food industry. Both cationic and zwitterionic surfactants are more for special use as they are more expensive to produce.
Because of their amphiphilic nature, surfactants absorb at the air-water or oil-water interface. At the interface, surfactants align themselves so that the hydrophobic part is in the air (or oil) and the hydrophilic part in water.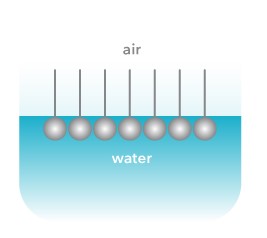
For simplicity, let’s consider only the air-water interface. The cohesive forces between the water molecules are very strong making the surface tension of water high. As surfactants absorb they break these interactions. The intermolecular forces between surfactant and water molecule are much lower than between two water molecules and thus surface tension will decrease. When the surfactant concentration is high, they form micelles. The point at which micelles are formed is called critical micelle concentration.
The main purpose of the surfactants is to decrease the surface and interfacial tension and stabilize the interface. Without surfactants washing laundry would be difficult and many food products like mayonnaise and ice cream would not exist. Thus optimization of surfactants for different applications is highly important and surface and interfacial tension measurements have a key role in it.
If you would like to read more about how surfactants are utilized in the industry, please download the overview below.
Surface tension plays an important role in Li-ion battery slurry optimization.
Surface tension plays an important role in the electroplating solution.
When measuring contact angles or making surface tension measurements with a pendant drop, selecting the correct tip or needle for your liquid is crucial.
The surface tension of water is about 72 mN/m at room temperature which is one of the highest surface tension for liquid.
Surface tension is a quantitative measure that can be correlated with a solution’s ability to remove dirt.
Surface tension and wettability are important physical properties that play a significant role in the effectiveness of agrochemicals.
Explains three different methods to measure surface tension.
The blog post describes what is surface tension and why is surface tension important in many industrial processes
The method to calculate the surface tension component is defined in ISO 19403-5:2017 standard.
