Exploring surface sensitive technologies, you often encounter the real-time method of QCM. This three-letter abbreviation is short for Quartz Crystal Microbalance, which essentially means that this is a balance for very small masses. If you have browsed the instrumentation area a bit more, you may have noticed that there are several versions of QCMs, such as QCM-D. So, what is the difference between QCM and the four-letter version where a D is added at the end?
Unlike traditional QCM, the QCM with an added D measures an additional parameter, namely the dissipation factor. The dissipation factor provides real time information about the softness and rigidity, also called the viscoelasticity, of the molecular layers on the sensor surface. This means that QCM-D provides more information about the system under study than traditional QCM.
So, QCM-D measures dissipation, but what is this parameter, what does it consist of and what does it mean? Sometimes dissipation is referred to as damping of the oscillating sensor. The strict definition of dissipation is the sum of all energy losses in the system per oscillation cycle. Using an equation to describe it, it can be written as 1/Q, where Q is the so-called quality factor of the oscillator used. Another way to define dissipation is the energy dissipated per oscillation, divided by the total energy stored in the system.
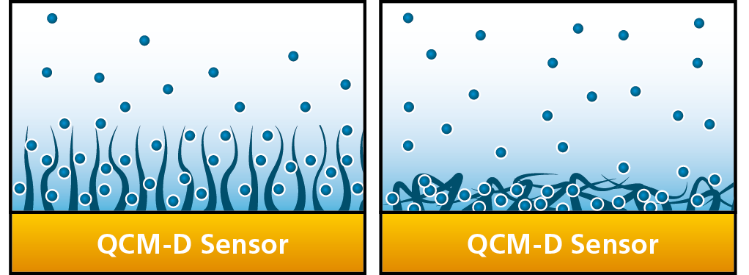
So much for the definition, but how is this related to an actual experiment? As already mentioned, the dissipation correlates to the softness of the layers on the sensor surface. A rigid material deposited on the sensor surface will follow the sensor oscillation without being deformed during the oscillatory motion. This means that a rigid material will not dampen the oscillation, and, in this situation, the measured dissipation will be low. On the other hand, a soft film will not be able to follow the sensor oscillations entirely. This means that it will be deformed during the measurement. The soft film will dampen the oscillation and result in high dissipation. Consequently, the dissipation parameter offers a real-time characterization of the softness or viscoelasticity of films deposited on the sensor surface, and it makes it possible to monitor conformational changes.

The possibility to collect more information about the system under study is of course positive, but is the dissipation information really needed?
There are three important aspects of the dissipation parameter:
Going into more detail about the first important aspect, the qualitative insight of the time resolved behavior of the system under study, the dissipation parameter will complement the information from the collected resonance frequency. Together the two captured parameters f and D will paint a picture of how molecules arrange themselves on the surface and how the arrangement varies with time; the frequency response of a QCM sensor reflects the amount of mass coupled to the surface, including the mass of solvent which is trapped in the molecular layer.
The dissipation reflects the softness of the film. Monitoring these two parameters as a function of time makes it possible to detect for example if molecules are lying flat on the surface (rigid film and low level of hydration) or if they are in a stretched configuration (soft film and high level of hydration), as well as if there are rearrangements such as for example swelling (from flat to stretched) or collapse (from stretched to flat), see fig 1.
To summarize, the dissipation parameter offers three important benefits over traditional QCM:
To get more out of your experiments, you can use the information that the dissipation parameter provides.
Download the white paper to read more about the differences between QCM-D and other QCMs.
Editor’s note: This post was originally published in November 2017 and has been updated
Read about what determines the sensing depth of the QCM-D technology and get examples of typical values
Register for the webinar
Read the guidelines on how to decide which QCM instrument will be the most suitable for your needs
Read about how protein adsorption at various surface and solution conditions quickly can be measured
Read about what single-harmonic and multi-harmonic QCM-D means and what the difference is between these instruments.
Read about how QSense QCM-D analysis is used as a powerful tool to investigate protein-lipid nanoparticles binding affinity
Learn about the difference between the theoretical QCM sensitivity and the sensitivity which is relevant in a measurement situation.
Read about the piezoelectric effect and how piezoelectricity arises
Learn more about QSense 4th generation QCM-D platform which provides a sharper tool in the scientist toolbox and simplifies data interpretation.
Fredrik is a Senior Application Scientist at Biolin Scientific. After his Master of Science in Biosensors- and Microsystems technology he has worked with technology and application development in as diverse fields as electroporation, multivariate gas sensing, drug screening and surface science.
